When discussing calcium chloride (CaCl₂), solubility is one of its most important properties. Its ability to dissolve quickly and completely in water makes it widely used in industrial, oilfield, food, and environmental applications.
Understanding calcium chloride solubility helps improve performance, safety, and cost control.
Why Calcium Chloride Is Highly Soluble
Calcium chloride is an ionic compound made of calcium and chlorine. When added to water, it dissolves and separates into ions:
CaCl₂ (s) → Ca²⁺ (aq) + 2Cl⁻ (aq)
This process is exothermic, meaning it releases heat. That is why the solution often feels warm after dissolving.
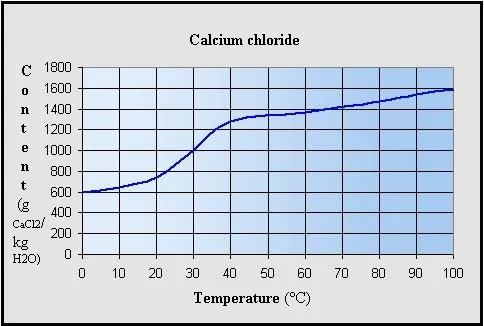
At 20°C, about 74 g of calcium chloride dissolves in 100 mL of water.
At 100°C, solubility increases to about 159 g per 100 mL.
Solubility increases as temperature rises.
Factors Affecting Calcium Chloride Solubility
Several factors influence dissolution:
- Temperature – Higher temperature increases solubility
- Concentration – Saturation limits dissolution
- Presence of other ions – Seawater and brines can change solubility behavior
- Ionic strength – May lead to complex formation or precipitation
In marine or high-salinity systems, ion interaction can modify how calcium chloride behaves.
Hygroscopic Nature: Why Storage Matters
Calcium chloride is highly hygroscopic. It absorbs moisture from air and can even dissolve in the absorbed water.
This property makes it an excellent:
- Desiccant
- Moisture absorber
- Container drying agent
However, it must be stored in airtight packaging to prevent caking or liquefaction.
Industrial Applications Driven by Solubility
1. Oil & Gas Industry
Calcium chloride brines are used in drilling fluids to:
- Maintain wellbore stability
- Control formation pressure
- Achieve required fluid density
High solubility ensures stable and concentrated brine systems.
2. De-icing and Anti-icing
Calcium chloride is widely used for:
- Road de-icing
- Sidewalk safety
- Airport runway treatment
Because dissolution releases heat, it melts ice faster than many other salts. It also lowers the freezing point and reduces refreezing.

3. Food Industry
Used as:
- Firming agent in canned vegetables
- Calcium source in beverages
- Texture stabilizer
Its high solubility ensures even distribution in food systems.
4. Chemical Production
Calcium chloride is used to produce:
- Calcium carbonate
- Other calcium salts
- Precipitation-based compounds
Its solubility allows controlled chemical reactions in labs and industry.
Environmental Considerations
When used as a de-icing agent, dissolved calcium chloride can enter soil and water systems.
At normal levels, it is generally safe. However, high concentrations may:
- Change soil structure
- Affect plant growth
- Alter water chemistry
Responsible dosage control is important.
Why Solubility Matters for Your Application
Understanding calcium chloride solubility helps you:
- Select the right concentration
- Optimize performance
- Reduce waste
- Improve storage stability
- Control environmental impact
Choosing high-purity calcium chloride ensures predictable dissolution and stable results.
Conclusion
Calcium chloride solubility is the foundation of its versatility. From oilfield brines to de-icing, food processing, and moisture control, its ability to dissolve efficiently makes it a critical industrial chemical.
If you are sourcing calcium chloride for industrial, oilfield, or environmental applications, selecting the correct grade and concentration is essential.
Contact us today to discuss your calcium chloride requirements and receive technical support tailored to your project.